Contents |
|---|
Purpose
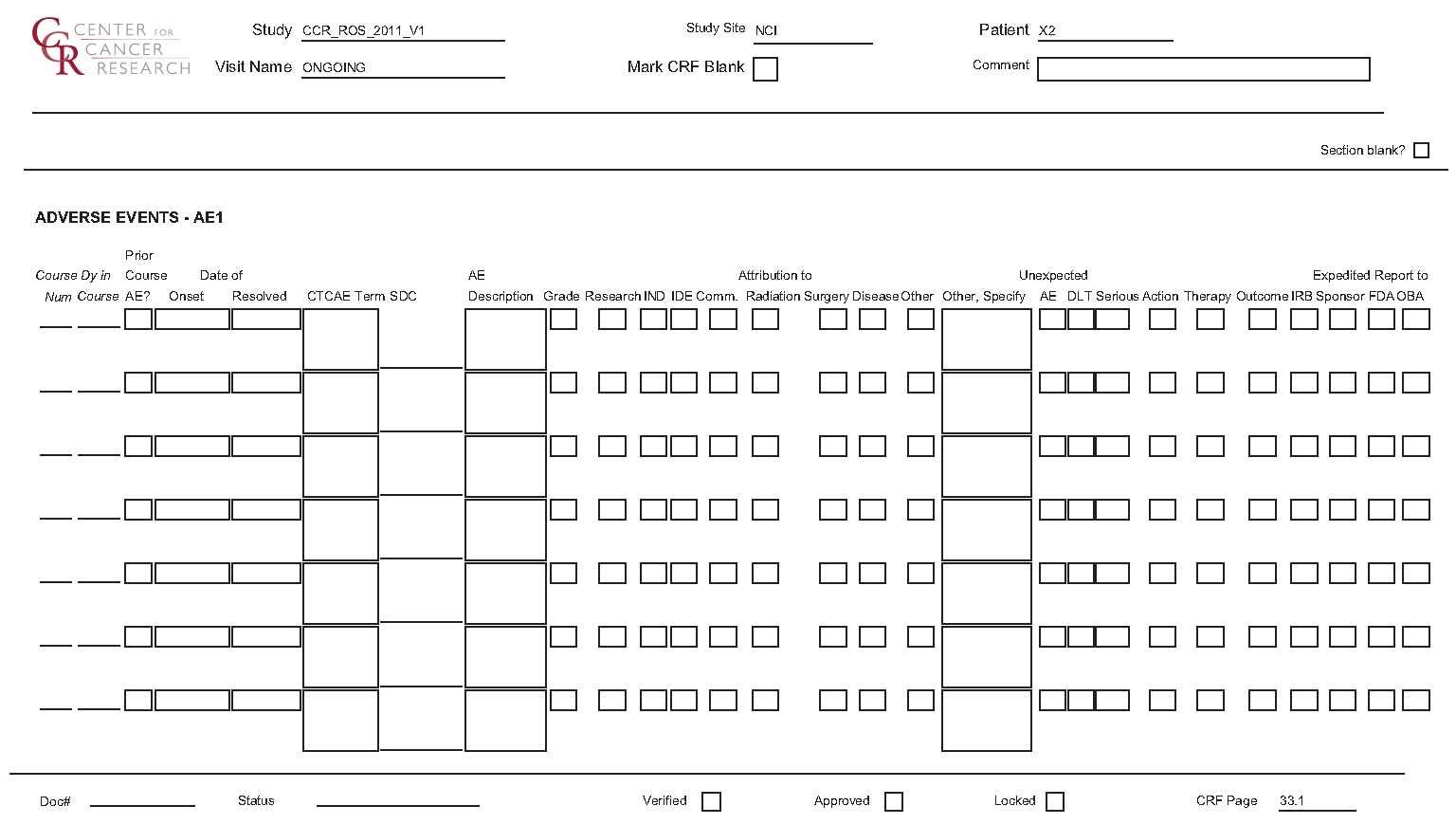
This eCRF is an ongoing form to capture all adverse events experienced by the patient regardless of the course.
An adverse event is any unfavorable or unintended sign, including abnormal laboratory findings, symptom or disease having been absent at baseline, or if present at baseline, appears to worsen, that has a temporal association with a medical treatment or procedure regardless of the relationship of the event to the medical treatment or procedure.
All adverse events will be coded using protocol specific version of NCI Common Terminology Criteria for Adverse Events (CTCAE) version. Every attempt to code the adverse event to a term using the standard terminology will be made before selecting the "other" term in a category.
Record all adverse events experienced by the patient, including laboratory abnormalities, regardless of relationship to the study medication.
An adverse event entry is composed of both the adverse event term plus the grade. Complete a separate row for each adverse event entry to be recorded using the appropriate adverse event term and the appropriate codes for "grade", "attribution(s)", "serious", "action", "therapy", and "outcome" in the respective column for each event.
If an adverse event has not been resolved, leave the Resolved Date blank. The Resolved Date can be filled at a later time when the adverse event is considered resolved. Resolution means a change in grade to a higher or lower grade.
If a patient died on study then the death adverse event onset and resolved dates should be the same.
How to record baseline symptoms that change, either improve or worsen:
If a pre-existing condition resolves, it does not need to be reported as an adverse event since it would have been already recorded on the Baseline Symptoms case report form. Enter the resolution date on the corresponding symptom entry on the Baseline Symptoms case report form improves, no entry is made on the AE eCRF. See BL eCRF for instructions.
If a pre-existing condition worsens (i.e.: the grade of the baseline symptom increases), that constitutes an adverse event entry which must be reported in full detail..
If a pre-existing condition improves without a resolution, do not enter as an Adverse Event. When it resolves, enter the resolution date on the corresponding symptom entry on the Baseline Symptoms case report form.
Adverse Events eCRF
The second screen shot is the portion to the right of the Grade The third screen shot is the portion to the right of the DLT
Field Descriptions and Instructions
Field Name | Description / Instructions | Format |
|---|---|---|
Visit Date | The Visit Date is optional on this case report form. Hit the "Tab" key to leave it empty and move to the Prior Course Adverse Event field. | DD-MMM-YYYY |
Course # (d) | Indicates the course number that this adverse event started in as derived from the course initiation start date.Late adverse event (For CTMS and CDS monitored studies, it means the adverse event observed after the date of off treatment) have no associated Course #. | 5 digits |
Day in Course (d) | Indicates the day since the beginning of course that this adverse event started as derived from the course initiation start date. | 5 digits |
Prior Course Adverse Event (c) | For adverse events that begin on the first day of a course, indicate if related to the prior course by entering:
| Use pick list. |
Date of Onset (m) | Enter the date of first observation of the adverse event and grade. If a patient died on study then start date and the resolve date for the death AE should be the same. | DD-MMM-YYYY |
Date Resolved | Enter the date of resolution of the adverse event and grade. Leave this field as well as the Outcome field blank if the adverse event is ongoing. | DD-MMM-YYYY |
CTC Term (m) | Using the pick list to select a CTCAE (Common Terminology Criteria for Adverse Events) Term. Note: This pick list does not show all the CTCAE Terms. User must type in a search criterion and then click on the ellipsis perform the search and display the resulting matched CTCAE Terms. Ex: type %ypo% to list all the terms that include the lower characters ‘ypo’ somewhere in the CTCAE Term. Note: Visit CTEP’s CTCAE webpage for latest version. | Use pick list. |
System Organ Class (d) | Broad classification of adverse events based on anatomy and/or pathophysiology. Within each category there is the adverse event term/description. Note: This field is derived from the selected CTC Term | 40 Characters |
Adverse Event Description | Enter a succinct clinical description of the adverse event. Note: This field is mandatory, unless thelor} CTCAE term is the same as the description (e.g. nausea, diarrhea). DO NOT enter raw data (i.e.: lab result). Use the term increase or decrease. | 100 characters(Only 33 characters are reported for CTMS monitored studies.) |
Grade (m) | Grade adverse events using Common Terminology Criteria for Adverse Events (CTCAE) version indicated in the protocol. Note: Note: Some grades are disallowed for some categories in the CTCAE. In the CTCAE tables this will be noted by the use of an em-dash "-". For example, Hair loss/Alopecia can only be graded as a 1 or 2, so grade 3, 4, and 5 do not exist and will be noted in the table with a "-" verses a description. If the protocol does not use either CTC or CTCAE, grade according to the following general criteria:
| Use pick list. |
Attribution to Research | Evaluate the adverse event's relationship to the subject's participation in the study, including study-related therapy and procedures. Select one of the following codes to record this evaluation:
| Use pick list. |
Attribution to IND (m) | Evaluate the adverse event's relationship to the investigational agent. Select one of the following codes to record this evaluation:
| Use pick list. |
Attribution to IDE | Evaluate the adverse event's relationship to the investigational device exemption. Select one of the following codes to record this evaluation:
| Use pick list. |
Attribution to Commercial | Evaluate the adverse event's relationship to the commercial agent. Select one of the following codes to record this evaluation:
| Use pick list. |
Attribution to Radiation | Evaluate the adverse event's relationship to the Radiation therapy. Select one of the following codes to record this evaluation:
| Use pick list. |
Attribution to Surgery | Evaluate the adverse event's relationship to the surgery. Select one of the following codes to record this evaluation:
| Use pick list. |
Attribution to Disease | Evaluate the adverse event's relationship to the disease. Select one of the following codes to record this evaluation:
| Use pick list. |
Attribution to Other | Evaluate the adverse event's relationship to other causes not listed above. Select one of the following codes to record this evaluation:
| Use pick list. |
Other, Specify | Enter an explanation when 'Attribute to Other' is selected. | 40 Characters |
Unexpected?(m) | Indicate if the adverse event is unexpected as defined by the NCI IRB, by entering:
| Use pick list. |
DLT (m) | Indicate if the adverse event is dose limiting, as defined in the protocol, by entering:
| Use pick list. |
Serious (m) | Indicate if the adverse event was a "serious" event by selecting from the following codes, as per the Code of Federal Regulations 21 Part 312. If multiple categories are applicable, select the worst.
| Use pick list. |
Action (m) | Indicate any changes made to the study regimen in response to the adverse event using the following codes. "Action" refers to the decision to reduce or continue the investigational medication.
| Use pick list. |
Therapy (m) | Indicate if additional therapy is required to treat the adverse event.
| Use pick list. |
Outcome | Select the final status of the patient when the adverse event is considered "resolved".
| Use pick list. |
Expedited Report to IRB (m) | Indicate if an expedited adverse event report was sent to IRB by entering:
| Use pick list. |
Expedited Report to Sponsor (m) | Indicate if an expedited adverse event report was sent to sponsor by entering:
| Use pick list. |
Expedited Report to FDA (m) | Indicate if an expedited adverse event report was sent to FDA by entering:
| Use pick list. |
Expedited Report to OBA (m) | Indicate if an expedited adverse event report was sent to OBA (Office of Biotechnology Activities) by entering:
| Use pick list |
Expedited Report to Manufacturer (m) | Indicate if an expedited adverse event report was sent to Manufacturer by entering:
| Use pick list |
Regarding the AE attribution fields:
For protocols that utilize the new system of AE attribution values (2 options only: “Adverse Event Unrelated” and “Adverse Event Related”), the following chart can be used to translate values in source documentation from the older system with 5 options to the new system. If the attribution in the source documentation is unclear the Data Manager should leave the attribution field in C3D/Rave blank until the source documentation is updated and clarified
Old System (numbers) | Old System | New System |
0 | Not Applicable | Adverse Event Unrelated |
1 | Unrelated | Adverse Event Unrelated |
2 | Unlikely | Adverse Event Unrelated |
3 | Possible | Adverse Event Related |
4 | Probable | Adverse Event Related |
5 | Definite | Adverse Event Related |
Legend: (d) derived field, (m) RDC mandatory, (c) for CTEP reporting only.
Validations
Code | Description | Resolutions |
|---|---|---|
AE01 | Date Resolved is before Date of Onset. | Correct either the Date of Onset or Date Resolve. |
AE03 | Two Adverse Event records have identical values for Date of Onset, CTC Term and Grade. | If duplicate, delete one of the records. If not, manually resolve the discrepancy. |
AE04, | Two Adverse Event records with the same CTC Term and/or Description have overlapping Date of Onset and Date Resolved ranges. | Correct the Onset and Resolution Dates for the Adverse Events in question or review/correct the CTC term/description |
AE08 | Adverse Event Description missing for some certain CTC terms that require a clinical description. | Enter the Adverse Event Description or review/correct the CTC term/description. |
AE09 | A Baseline Symptom exists with the same CTC term and Grade as the Adverse Event and the Baseline Symptom has not been resolved. | Verify the Baseline Symptom resolution date, the Adverse Event onset date, or CTC Term. |
AE10 | The CTC Term for the ongoing Adverse Event has a specified lab, but a lab record with lab date = AE onset date and lab grade = AE grade does not exist. | Verify that the Averse Event is supported by appropriate lab test result. |
AE11 | Adverse Event is resolved and there is no supporting lab test result. | Review Adverse Event and related lab test result and their grades. |
AE14, | The Adverse Event Date of Onset or Date Resolved is in the future. | Correct the Onset or Resolution Dates. No future dates should be recorded. |
AE16 | The Adverse Event Date of Onset is less than the first Course Start Date. | Correct the Adverse Event Date of Onset to be equal to or after the first Course Start Date. |
AE17 | The Adverse Event CTC Grade is invalid. | Enter a Grade that is permissible for the CTC Term. |
AE19 | Resolution date has been entered, but Outcome is not provided or vice-versa. | A Date Resolved must be accompanied by an Outcome and vice-versa. |
AE20 | Adverse Event is the cause of death but Grade is not 5-Fatal and/or Outcome is not 4-Died and/or Seriousness is not 3-Death. | Change the Adverse Event Grade, Outcome and Seriousness. |
AE21 | Prior Course checked 'Y', but there is no Course with a Start Date the same as the Adverse Event Onset Date. | Change the Adverse Onset Date, the Prior Course or the Course Initiation Start Date. |
AE22 | Adverse Event 'Attribute to Other' and 'Other, Specify' are not present together. | Enter 'Other, Specify' if 'Attribute to Other' is associated. |
AE23 | Adverse Event Attribution to Research is not the same as the highest Attribution to IND, IDE, Commercial, Surgery, and Radiation | Review all Attributions to make sure that Attribution to Research is same as the highest Attribution to IND, IDE, Commercial, Surgery and Radiation. |
Derivations
Code | Field Name | Description |
|---|---|---|
AE1002 | Course # | Course number is derived based on the course initiation start dates. |
AE1003 | Day in Course | Number of days since the beginning of the course is derived from the course initiation start date. |
AE1004 | CTC Category | Broad classification of the CTC Adverse Event Term derived from the pick list selection. |