General Background
Pteridine nucleoside analog probes are highly fluorescent as shown by the quantum yields listed below. Similarities in structure and size to native nucleosides make it possible to incorporate these probes into oligonucleotides using phosphoramidite chemistry and automated DNA synthesis. As a part of an oligonucleotide the probes are exquisitely sensitive to changes in structure as the oligonucleotide meets and reacts with other molecules. Changes are measured through fluorescence intensity, anisotropy, lifetimes, spectral shifts, and energy transfer.
With most bulky, linker attached probes, the degree to which they sense changes in the DNA structure is minimal. The unique aspect of the pteridines analogs is that they are incorporated into DNA through a deoxyribose moiety identical to that of native DNA using phosphoramidite chemistry (with no "linker-arm" attachment involved). This native-like linkage causes the probes to base stack with neighboring bases rendering them exquisitely sensitive to subtle changes that occur within the DNA. Changes in base stacking or base pairing in the vicinity of these probes are reflected by distinct changes in the fluorescence properties of the pteridine. These probes also have a favorable spectral position, with absorption near 350 nm which overlaps well with the emission spectrum of tryptophan thus permitting their use as energy transfer partners with tryptophan using fluorescence resonance energy transfer (FRET).
Sequence | Fluorophore Pairing Partner | T | A | C | G |
PTR22 | 6MAP | 53.5°C | 43.8°C | 42.8°C | 45.2°C |
Tm of native sequence is 57.8°C
This table shows the melting temperatures for oligonucleotides containing 6MAP paired with complementary strands with substitutions of T, A, C, or G as pairing partner for the fluorophore. 5'-gtg tgg aaa Ftc tct agc agt-3'. This demonstrates that the probe pairs most stably with T as would be expected for an adenine analog.
Probe | EX max | EM max | Q rel |
|---|---|---|---|
3-MI | 348 nm | 431 nm | 0.88 |
6-MI | 340 nm | 430 nm | 0.70 |
6-MAP | 330 nm | 435 nm | 0.39 |
Fluorescence properties of pteridine nucleoside analogs.
EX max Excitation wavelength maximum
EM max Emission wavelength maximum
Q * rel* Relative quantum yield
Monitoring Real time digestion of DNA
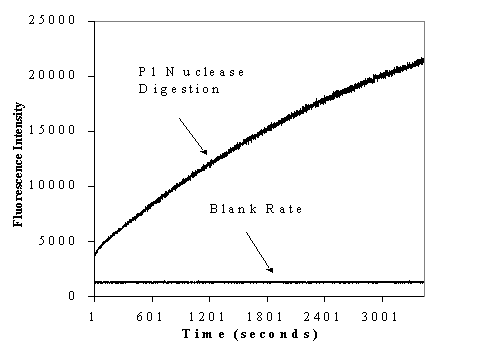
One way to make use of the "self-quenching" property of these probes is to use it to monitor events that change the base stacking interactions of an oligonucleotide. Fig. 2 shows how one can observe the fluorescence intensity changes that occur when a single strand containing a pteridine probe is digested using P1 nuclease.
The data shown above is generated by digesting a pteridine-containing oligonucleotide using P1 nuclease at 37 C while monitoring emission at 450 nm with excitation at 350 nm. Data was collected in real time. The blank rate shows an identical solution under identical conditions without P1 Nuclease. This is an example of how these probes can be used to monitor changes such as cleavage or digestion of DNA caused by action of a protein. |
Use as Hybridization Probes
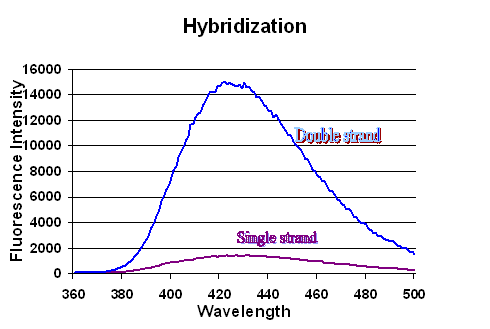
Another application of these probes takes advantage of quenching caused by base stacking. Upon annealing two complementary strands which do not contain base pairing partners for the fluorophores, an increase in fluorescence is generated because the probes are pushed out of the base stacking environment forming a single base bulge. The maximum amount of fluorescence increase generated varies depending on the base sequence surrounding the probe and the concentration.
Fluorescence emission scan of single and double strands of the same 3MI-containing sequence. Emission scan of equimolar concentrations of 3MI in a single strand, 5'-atc ctg gga tta aat aaF aat agt aag aat gta tag ccc tac-3', and of 3MI in the same sequence annealed to its complementary strand, 5'-atc ctg gga tta aat aaF aat agt aag aat gta tag ccc tac-3': 5'-tga ggg cta tac att ctt act att tta ttt aat ccc agg at-3'. The 3MI in the double strand environment does not have a base pairing partner. The scans shown in this figure are displaying an increase of approximately 7.5 fold for that particular oligonucleotide at that concentration. |
Structural probing of a sequence using 3-MI and 6-MI
Recent experiments published in the article (Biochemistry, 2009) have revealed complexity in DNA conformation using the two guanine analogs, 3-MI and 6-MI. Results confirm that 6-MI is preferred for anisotropy measurements because it more closely reports on the motion of the strand. Quasi static self quenching is also revealed as a large component of quenching for the pteridine probes and most likely for other base stacking analogs. |